|
We'll see that these three states in the triplet don't have exactly the same energy and only one is the * true ground state *. 9 microstates constitute the same state (and energy) called ³P, in which the total magnetic momentum can assume three different values (0, 1, 2). The two 2p electrons can be placed in 15 different combinations in the three available 2p orbitals ( #2p_x#, #2p_y#, #2p_z# corresponding to orbital magnetic momentum -1, 0, +1) and with electron spins up (↑ = +½) or down (↓ = -½). The most stable electronic configuration DOES NOT correspond to the ground state you are asking about (these are two completely different concepts), because there are usually several different states, with different energies, corresponding to the same electronic configuration, but ONLY ONE is the ground state, the one with the lowest energy.įor example, a simple six electron atom as the carbon atom has the most stable electronic configuration represented by 1s² 2s² 2p². 
In general, to that state correspond many different microstates having exactly the same energy, but different occupation and spin of the valence electrons in the partially filled orbitals. That value corresponds to the minimum energy that you should furnish to extract the less bounded electron from the atom, that is the first "ionization energy", and that energy is the same of the GROUND STATE. 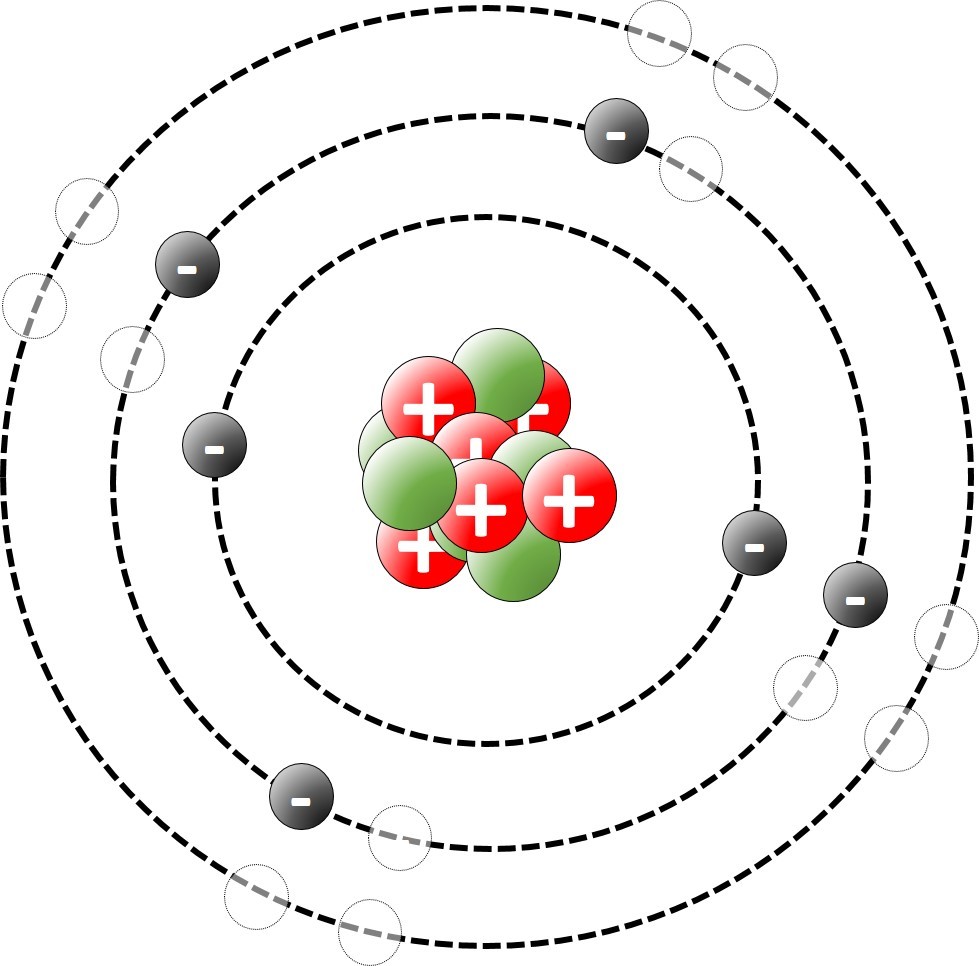
It can be experimentally recognized from the absorption spectra of the atomized element in the gas phase: there is a series of lines, in the spectrum, whose frequency reaches a maximum value. 
The ground state is the state that is occupied by the most part of the atoms of the same element at room temperature, because it is lower in energy.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
